3D Printing Stem Cells
Latest News
June 14, 2016
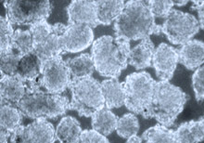
Nano Dimension, a company more commonly associated with 3D printed electronic components, has teamed with Israeli stem cell research company Accellta to test a 3D bioprinter that uses stem cells.
The two companies tested the printer in May and plan to launch a joint venture focused on stem cell printing, and ultimately bioprinting human organs and tissues.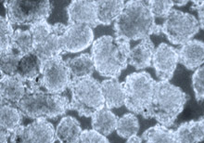
“3D printing of living cells is a technology that is already playing a significant role in medical research, but in order to reach its full potential, for the field to evolve further, there is a need to improve printing speeds, print resolution, cell control and viability as well as cell availability and bio-ink technologies,” said Amit Dror, CEO of Nano Dimension. “By combining our high speed, high precision inkjet capabilities with Accellta’s stem cell suspension technologies and induced differentiation capabilities led by a world-renown group of experienced engineers and scientists, we can enable 3D printing at high resolution and high volumes.”
The solution tested in the feasibility study leverages Accellta’s suspension-based cell culturing systems, which can produce billions of stem cells per batch. With large quantities of quality cells, the companies could potentially print larger and more complex tissues and organs.
“Accellta and Nano Dimension have joined forces in this initial trial to evaluate and adapt the joint potential of our technologies. We hope and believe that this will bring the mutual capabilities and know-how of both companies to create 3D bioprinting solutions that combine a high precision, high-throughput printer with dedicated bio-ink technologies, derived from stem cells,” said Dr. Itzchak Angel, Chairman and CEO of Accellta. “By enabling high precision 3D bioprinting and differentiation of stem cells into required tissues, our combined technologies have the potential to enable vast areas of development. We are very excited about these initial results and what the future holds.”
In June, Nano Dimension filed a patent application in the U.S. that covers aspects of 3D printing human tissues and organs using stem cells and inkjet technology. It also includes converting MRI and CT scans and resulting images to create 3D structures for bioprinting.
Source: Nano Dimension
Subscribe to our FREE magazine, FREE email newsletters or both!
Latest News
About the Author
Brian Albright is the editorial director of Digital Engineering. Contact him at [email protected].
Follow DE





