Stent Simulation: Big Complications in a Small Package
Stent simulation requires training to develop engineering skills and workflows. Software providers are stepping in to provide it to their medical customers.
March 1, 2014
By Jamie J. Gooch
Imagine designing a flat structure—the thinner, the better—that can be inserted into a pipeline from afar, and then be expanded into a tube-shaped scaffold that will support the pipeline’s deteriorating walls. Now imagine that those walls are constantly moving, requiring the scaffold to compress and expand again and again.
Such form, fit and functionality requirements would be a challenge for any engineer. But when the pipeline is an artery, the scaffold a tiny stent, and the engineer a medical researcher—the complications multiply.
Talking the Talk
Consider the challenge faced by Shannon Gott, a Ph.D. candidate at the University of California, Riverside. Gott is researching a new type of stent that she expects to cause less damage to artery wall linings, which can cause tissue to build up. That buildup, called restenosis, narrows the artery, defeating the purpose of the stent. The other big risk is thrombosis, or blood clotting, sometimes tied to drug-eluting stents that release therapeutic agents to reduce restenosis.
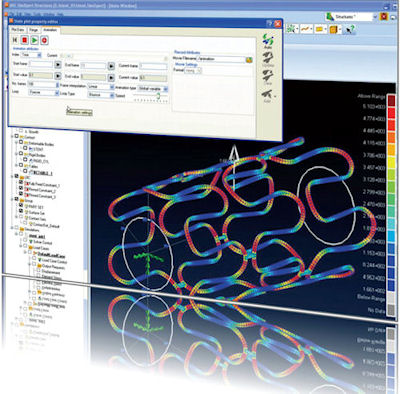 MSC’s SimXpert (shown here) integrates multi-discipline analysis capabilities that can be used to simulate stents from pre- to post-processing. |
“We’re trying to address some of the problems that stents currently have by nanopatterning the surface of stents to get the desired cell response from vascular cells, and promote healing that way,” she said at the Siemens PLM Software NX CAE Symposium last year. “Because we’re trying to nanopattern the surface, we need to develop and refine titanium micromachining techniques, and we’re coming up with a new planar design.”
Restenoisis, thrombosis, therapeutic agents, nanopatterning? The language barrier alone between medical terms and simulation terms—such as oscillating stress vs. mean stress, contact non-linearity or even computational fluid dynamics (CFD)—can be a barrier to innovation.
Gott received a helping hand from Andrew Jabola, an application engineer at software reseller and service provider Saratech who, in his own words, “analyzed and correlated current planar stent designs against physical test data, and optimized future designs using FEA (finite element analysis)” capabilities in Siemens NX computer-aided engineering software.
That need for a translator between the medical and simulation analysis fields, not just of industry jargon but of specialized knowledge, is an opportunity for many engineering service providers, software resellers and developers.
“I think that’s part of the reason my position was created: to have someone who can learn the language of biomedical folks,” says Kristian Debus, director of Life Sciences at CD-adapco. “My task is to get into the biomedical community, plus to internally educate our own people.”
Multiple Paths to Innovation
Debus says many CD-adapco customers he works with know the biomedical side, but not the engineering side. His challenge is to get everyone up to speed using CD-adapco’s STAR-CCM+ analysis environment. The company provides in-house, on-site and on-demand training, as well as service contracts where analysts will perform the simulation for the client.
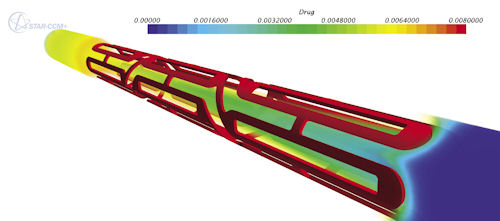 CD-ADAPCO’s: STAR-CCM+ can be used to simulate flow and pressure through stents while automating many time-consuming aspects of the workflow. |
“In a service contract, our goal is to transfer the information and knowledge to them, then sell them a software license so they can do it themselves—with the help of a dedicated support engineer they can call on as needed,” he adds.
For example, professors Alan Edelman, Ph.D., and Caroline O’Brien Ph.D., at the Massachusetts Institute of Technology (MIT) have mechanical and biomedical engineers on their research team. At the Edelman Lab, they are working to determine how tissue responds to implanted devices, including stents, and how their design influences that response.
“Within our current work, we integrate computational modeling with state-of-the-art intravascular imaging to extract real-world flow domains,” Edelman explains. “The simulation of the flow fields in these image-derived geometries is notoriously difficult, however ... such a simulation has been made possible through CD-adapco.”
For other customers who don’t have the diverse staffing resources available at MIT, that hand-off to independent analysis is still possible. Simulation software is becoming easier to use and the necessary simulations are better understood, according to Derek Barkey, senior manager of Consulting at MSC Software.
“What used to be a fairly complex analysis is now actually pretty routine,” he says. Still, many of MSC’s life sciences customers would rather leave the analysis to the experts.
“Usually they have no idea about the simulation work,” he says. “It’s not a core competency they want to develop. Usually they’re pretty well acquainted with FDA (U.S. Food and Drug Administration) requirements and have read their guidelines and know what needs to be done.”
On the flip side, software developers and engineering service providers also train engineers in the specifics of stents. For example, Dassault Systemes SIMULIA is offering “Modeling Stents Using Abaqus,” a two-day course designed to help engineers create geometry for modeling stents; choose the proper element type; perform static, implicit and explicit dynamics analyses; and more. The course is next being offered May 29-30 in Warwick, RI.
Opportunities Abound
While life sciences researchers are accustomed to long lead times associated with decade-long clinical trials and the peer review process, they are often surprised by the time it takes to set up and run a simulation, Debus says. The need for an efficient workflow and the computing horsepower required for complex simulation runs are the primary speed bumps.
“You can do lot on desktop these days, but our prime customers are on clusters,” he says. “Big models require horsepower. Others are using low-level simulation, but to go really deep takes an investment in hardware and manpower.”
Biomedical researchers aren’t like automotive and aerospace customers, he adds. They don’t always have the system in place to support the hardware and software they need.
An example can be found at MIT, where integration with Simpleware Ltd.’s ScanIP image processing software was causing a bottleneck. Edelman says the staff at CD-adapco facilitated a collaboration “to create an efficient workflow taking intravascular images through to flow simulation.”
As the stent research landscape shows, the software training, hardware selection and design cycle optimization needs of the life sciences community present many opportunities for service providers. The key to taking advantage of those opportunities is in ensuring you have the necessary expertise in the specifics of various biomedical fields, not just in various engineering disciplines.
Jamie Gooch is managing editor of Desktop Engineering. Contact him via [email protected].
More Info
Subscribe to our FREE magazine, FREE email newsletters or both!
About the Author
Jamie Gooch is the former editorial director of Digital Engineering.
Follow DE





