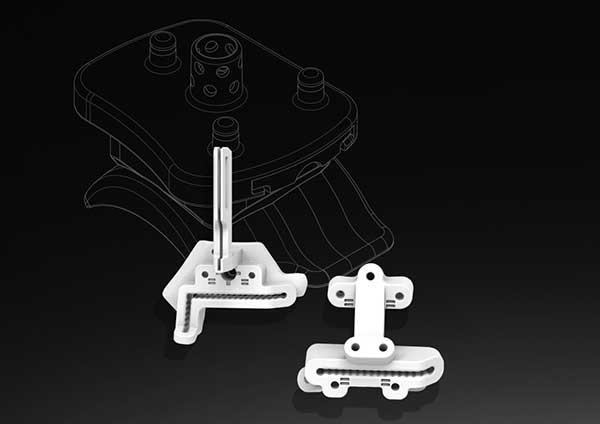
3D Systems has announced the Food and Drug Administration (FDA) has provided 510(k) clearance for the Vantage Ankle PSI, its patient-specific total ankle surgical planning and 3D printed instruments. The product includes pre-surgical planning and a patient-specific 3D-printed instrument set that guides resections in the tibia and talus for total ankle replacement surgery using Exactech’s Vantage Total Ankle System.
Vantage Ankle PSI increases operating room efficiency, reliability and improves soft-tissue preservation around the joint. This innovation is a result of the collaboration between 3D Systems and Exactech, a developer and producer of innovative implants, instrumentation and computer-assisted technologies for joint replacement surgery.
Patient-specific orthopaedic instruments are an enabling technology that help surgeons prepare the skeletal anatomy to receive an implant. The Vantage Ankle PSI product, a solution to facilitate direct patient-specific osteotomies in the ankle, is designed to increase surgical efficiency by allowing the surgeon to reduce the number of steps required to prepare the anatomy with a patient-matched 3D-printed instrument set.
Features unique to the product include a large footprint that helps to reliably seat the guide on the bone anatomy, improved visibility to alignment and a corrugated design on the cutting slots that aid surgical irrigation. This is complemented by soft tissue offsets designed to preserve the periosteum, the outer fibrous layer of the bone that aids in its healing and recovery.
“3D Systems was founded on a spirit of innovation, and we are constantly looking for opportunities to expand the applications that can be addressed with our VSP surgical planning solutions to benefit the medical community,” says Menno Ellis, EVP, healthcare solutions, 3D Systems. “Through our collaboration with Exactech, we drew upon our collective expertise in orthopaedics to develop an end-to-end solution for total ankle replacements that is unique to the market.
"The combination of pre-surgical planning and 3D-printed, patient-specific instruments allows the surgeon to visualize the patient anatomy and surgical approach in three dimensions, and then perform the surgery more efficiently with improved surgical outcomes,” Ellis adds.
“Our collaboration with 3D Systems represents a meaningful advancement that will accelerate the success of our Vantage Ankle prosthesis,” says Exactech CEO Darin Johnson. “Their expertise in surgical planning, as well as medical device design, regulatory, and manufacturing was invaluable in developing this patient-specific solution. The Exactech team is eager to bring this solution to market to simplify ankle arthroplasty’s surgical technique and enhance the patient and surgeon experience.”
3D Systems and Exactech have entered into a distribution agreement for the Vantage Ankle PSI product offered exclusively with Exactech’s Vantage Total Ankle System. The Vantage Ankle PSI is currently in pilot launch with full market availability expected in late 2021. Interested surgeons can contact their Exactech sales representative directly, visit Exactech’s website or join a live webinar on December 1, 2020 to learn more about this product.
Sources: Press materials received from the company and additional information gleaned from the company’s website.


From digitization, design and simulation through manufacturing, inspection and management, our comprehensive portfolio of technologies provides a seamless, customizable workflow designed to optimize products and processes while accelerating…

Save Time and Reduce Costs with NoSupports™ Metal Printing
Metal 3D printing is a high energy process in which it is possible for unmanaged heat and stress to build up and impact part quality, this guide presents a step-by-step methodology for how to apply NoSupports to a hot end section of a larger turbomachinery part.
DE's editors contribute news and new product announcements to Digital Engineering. Press releases may be sent to them via [email protected].
Follow DE
Join over 90,000 engineering professionals who get fresh engineering news as soon as it is published.


