Latest News
February 8, 2018
The FDA’s decision to encourage new methods of modeling and simulation to accelerate the pace of innovation in life sciences is likely to have far-reaching consequences for product design, development and regulatory approval in that industry. Digital design tools have emerged as a key contributor to a manufacturer’s success in this environment.
 Mahesh Kailasam, Ph.D.
Mahesh Kailasam, Ph.D.Thornton Tomasetti’s Applied Science practice has an almost 70-year history of pioneering and engineering practical solutions to various challenges in multiple industries by performing research, modeling, physical testing and analysis for clients around the globe. The company has now expanded its practice into life sciences, tapping Mahesh Kailasam, Ph.D., to lead the initiative from the firm’s Silicon Valley office. Dr. Kailasam has more than 20 years of experience in the deployment of modeling and analysis techniques in emerging technology sectors.
He most recently led the commercialization of the technology developed in the Living Heart Project, and previously worked on pioneering modeling and simulation-based approaches for exploration and production methodologies in oil and gas production. As vice president of Thornton Tomasetti, Dr. Kailasam will leverage Applied Science’s expertise in modeling and simulation to accelerate innovation in the worlds of high tech and life sciences, and the growing intersection of the two.
Dr. Kailasam was recently interviewed about the role of digital design tools in meeting the FDA’s challenge to speed product development in life sciences. He was joined by Steven M. Levine, Ph.D., senior director of life sciences at Dassault Systèmes SIMULIA, and Thomas Z. Scarangello, chairman and chief executive officer of Thornton Tomasetti.
Q: What are the most acute challenges faced by life sciences/medical device developers/manufacturers today?
Mahesh Kailasam, Ph.D. (MK): Our clients face three primary challenges today. The first is the challenge of cost—how do you develop new products in a cost-effective manner, especially as the costs of medical treatment are rising everywhere? The second challenge is to make sure that whatever solutions are developed are applicable for the targeted patient population so that treatments are effective. Historically, solutions have been developed in a generic sense, but applied in an individualized sense using experience and observation. The challenge we face now is getting to a point where devices and treatments are personalized to an individual’s characteristics—while keeping the first challenge of cost in mind—so that the treatment works as intended both in the near-term and in the longer term. The third challenge is time, the need to develop solutions in a timely manner.
When you consider these all together, it is clear that the heavy reliance on traditional processes, including bench-top experiments, animal testing and typical clinical studies, is just not suited for the challenges the industry is facing right now. Digital technologies will be key to accelerate the transformation that is needed.
 Steven M. Levine, Ph.D.
Steven M. Levine, Ph.D.Steven M. Levine, Ph.D. (SL): What we are hearing is that the single biggest way to reduce the skyrocketing costs in the healthcare industry is to lower the volume of post-treatment care. This has two components, (1) getting the treatment right the first time, and (2) shortening recovery time with less invasive treatments. The former often is characterized as “precision medicine,” but it basically means that we need better ways to analyze a given condition and select the best treatment. The use of digital technologies will be transformational in this, from using real-world data and in silico models to develop better physiological models of patients, to conducting virtual treatments to optimize outcomes. The latter involves providing physicians with a more targeted approach in situations where they have less ability to see what is happening inside the body. Once again, virtual reality and realistic digital representations of the patient and procedure are critical to make this happen. Also, incorporating real-world behavior as part of the diagnostic or follow-up can dramatically improve success while achieving cost-saving goals.
Q: What is the FDA doing to support, yet regulate, the industry?
MK: The FDA has been encouraging the adoption of new modeling and simulation technologies for effectively evaluating different solutions and accelerating the pace of innovation. Simultaneously, in order to make sure that such adoption is done in a consistent manner, there are emerging guidelines, such as for verification and validation, to ensure that this is done in a systematic and controlled manner rather than an arbitrary way. In addition, programs such as MDDT (Medical Device Development Tools) are helping the industry develop virtual human and animal models to evaluate medical devices with greater confidence than before.
SL: I understand the challenge the FDA has in maintaining their need to oversee the introduction of safe medical devices, while at the same time accepting responsibility to help lower the speed and cost barriers without compromising safety. To meet this challenge, the FDA has invested, through internal R&D as well as extensive collaborations, in understanding new methods that could achieve both goals. They have evaluated the various sources of evidence, animal models, clinical trials and computational models and concluded that, in many instances, as much as 50% of the time the computational models could be a better source.
As such, they are actively working internally and through collaborations with organizations such as the Medical Device Innovation Forum (MDIC) and projects such as the Living Heart Project (LHP) toward a future where half or more of the data submitted for regulatory approval comes from computer modeling, virtual patients or virtual clinical trials. Moreover, they are publicly sharing this mission, publishing guidelines such as the V&V40, and encouraging what they have called a “Simulation Revolution” in medical devices.
 Eye-optimal device.
Eye-optimal device.Q: Why are digital tools essential at this point?
MK: Digital tools allow us to virtually try out multiple solutions to any challenge and do it all efficiently. This is certainly relevant at the earlier stages of design evaluation via virtualized benchtop tests, where companies ranging from the largest medical device makers to hundreds of startups can use simulations to effectively zoom in on designs that have the most promise. These tools are also very relevant, and perhaps more valuable, in later stages where digital models of organs like the heart or skin can be used to assess effectiveness of devices in virtual populations and tailor the solutions to different population segments. Information from such virtual studies will allow clinical studies to be more effectively designed, giving device developers greater confidence in their outcomes; this will lead toward virtual clinical trials in the near future.
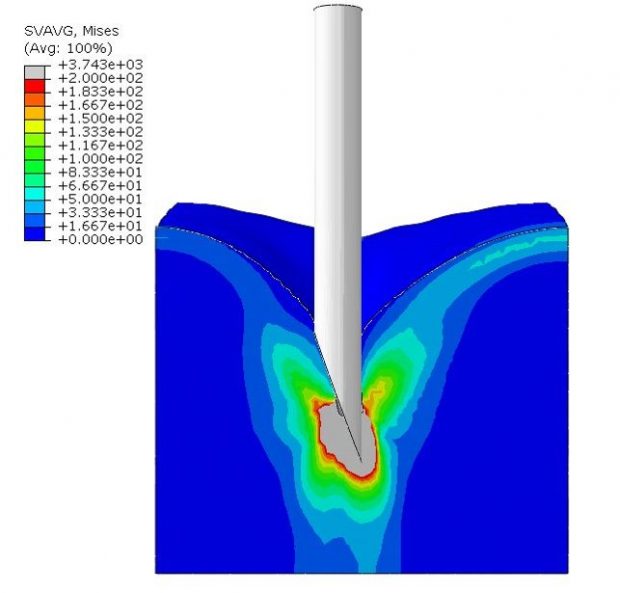 Skin needle penetration.
Skin needle penetration.
 Thomas Z. Scarangello
Thomas Z. ScarangelloThomas Scarangello (TS): Our firm has a long history of using digital tools for modeling and analyzing large and complex systems, including the effects of underwater shock on submarines and ships, ground-borne seismic waves and shock on structures, and environmental loading on structures like supertall skyscrapers, stadiums and arenas. These tools have created tremendous efficiencies and have been proven vital in speeding the pace of innovation and the delivery of new solutions for our clients in these industries, and they will play the same role in helping our life sciences clients accelerate their success as well. Many of the methods we have been using are directly applicable to life sciences, particularly when you relate the underlying physics—whether they are structural, thermal, CFD and so on—to the problems at hand. Digital tools are perfect for capturing the interactions between different aspects of any complex system, whether it is the behavior of submarines underwater or stents experiencing blood flow inside vascular systems.
Q: What are some examples of successful projects already being achieved in the industry?
SL: Five years ago you would have been hard-pressed to identify more than a few examples where simulation was on the critical path to commercial success of medical products. However, there were several showcase examples, such as cardiovascular stents where simulated fatigue prediction has become necessary to ensure not only the safe lifetime of a new design, but also the regulatory approval of the FDA. Today, nearly every device can be MRI-safety certified virtually, and the FDA even has a formal program called MDDT (Medical Device Diagnostic Tools) to precertify computational methods that can speed regulatory approval. I also believe the FDA has sent a clear message to the device community when they joined the LHP to lend their support to introduce this technology into the regulatory process.
More importantly, the LHP, now entering its fourth year, has demonstrated that physics-based simulation, once the exclusive domain of mechanical devices, is equally applicable to biological systems such as the human heart. Using a consensus methodology among the now 100+ members, models and methods to virtually design and test new devices have been developed. These methods offer the potential to test and refine new device designs in a fraction of the time and cost of current methods that are based on a combination of bench and animal testing, and over time to be more predictive of clinical performance. Using the LHP as a basis, Dassault Systèmes is now working on models for other medically important body systems such as the brain, knee, spine etc.
MK: Digital tools are being used successfully in many areas, including where personalization is important. As an example, digital modeling and analysis were already being used to develop high-quality implants, but now many solutions (knee or hip replacements among them) are being designed to match an individual’s lifestyle choices, such as athleticism, as well as his or her physical characteristics, on a far more granular level.
In addition, these digital tools also are enabling production of high-quality, patient-customized implants using additive manufacturing (3D printing). These solutions are not only being offered by large medical device companies but also by a whole cadre of smaller startups, including several that are able to recommend strategies for surgery and treatment based on simulated estimates of post-treatment outcomes.
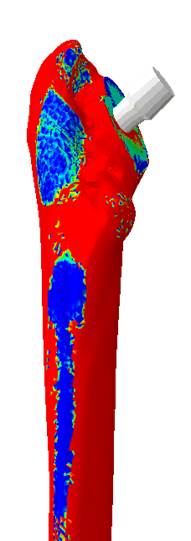 Hip personalized bone implant stress.
Hip personalized bone implant stress.Similarly, in other domains such as vasculature and blood flow, digital tools are being used to develop models of the vasculature from imaging data, which can then be used for a variety of applications—ranging from 3D printing of realistic blood vessel network models to simulating the behavior of devices inside these models, and to even assessing various disease conditions that may hinder blood flow or risk the integrity of the vasculature.
Q: Why has Thornton Tomasetti (TT) decided to expand its life sciences capabilities at this time?
TS: Our decades of leadership in virtual modeling and simulation to enhance designs translate perfectly into the biomedical field, especially as the FDA and other agencies are also promoting simulation as a means of safely accelerating approval cycles. Many of the tools and modeling capabilities that TT has been developing and honing since 1949 are at the cutting edge of some of the principles that are now being promoted by the FDA and increasingly being adopted by the life sciences community. For example, I expect that our experience with stochastic and probabilistic assessments of various types of events will allow us to offer methods that are quite relevant to life sciences, considering the variations in population or disease characteristics.
MK: Yes, our development and use of vibration and piezoelectric methods has played a key role in improving ultrasonic imaging solutions and is already being used by leading medical imaging companies. Another example is the development and pioneering adoption of constitutive models for soils, which at first glance doesn’t appear relevant to life sciences until you realize that the same constitutive models are used to simulate the manufacturing of pharmaceutical tablets. Overall, the firm’s commitment to driving innovation is at the heart of all we do, and is directly translatable to what we are able to bring to the life sciences arena.
Q: Mahesh, tell us a bit about your background and why you’ve joined TT Applied Science.
MK: I’ve spent the last two decades promoting the adoption of modeling and analysis techniques in a variety of industries, including life sciences, high tech and energy sectors, and helping identify and develop new methods. I was doing all this from the point of view of getting customers to adopt specific digital tools and less from the point of view of actually helping them use these tools to solve their pressing challenges.
Today, at Applied Science, my priority is to solve our clients’ problems and to transfer the methodologies to them for future reuse. In fact, the primary mission at the core of all of TT’s work is to embrace and solve the toughest challenges of our clients and to make lasting contributions for their benefit. Working with a team of gifted engineers, I am now able to show our clients how digital tools can solve some of the challenges they’ve been facing for years. The opportunity to work with clients in this manner, and TT leadership support to build an even stronger team to expand our reach in life sciences, are the main reasons I joined the company.
Q: Why is TT Applied Science particularly qualified to contribute in this arena, and what are your capabilities?
MK: TT Applied Science has been using digital modeling and simulation for longer than most companies—it’s in the DNA of our firm. We’ve used modeling and simulation to solve problems and analyze complex systems in a number of other industries, and there are many similarities between TT’s historic uses of modeling and simulation to the current needs of the life-sciences world. We have also been active with various ASME committees for computational modeling, such as V&V 10 (Solid Mechanics), V&V 20 (Fluid Mechanics and Heat Transfer), V&V 50 (Advanced Manufacturing, covering additive manufacturing) and more recently have started engaging with V&V 40 (Medical Devices) as well.
In terms of capabilities, we have expertise with a wide variety of physics modeling solutions, covering everything from solids and fluids to thermal and electromagnetic simulations. We also have the capabilities to research and develop new material models, develop bespoke test fixtures and carry out physical tests to help with validation, and to develop and automate new or existing methodologies, such as for probabilistic assessments or optimization, for our clients.
We have expanded and are continuing to grow our team of engineers with specialized skills in bioengineering. This allows us to provide virtual human modeling and simulation solutions covering a range of devices and organs, using both already available virtual models or, when needed, starting from imaging data to develop simulation-ready virtual human models, and then performing needed simulations. After the simulations and assessments are completed, we help our clients prepare thorough reports for submission to the FDA or other regulatory bodies, including necessary evidence of the efficacy and validity of the simulations. If they are interested, we also provide them with effective visualization solutions such as realistic rendering or VR approaches.
We have established strong partnerships with companies like Dassault Systèmes and others to leverage the best available digital tools and bring to bear our expertise on addressing our client’s individual challenges.
Q: Mahesh, what directions do you see life sciences product development taking in the future, and what are your goals for your own division?
MK: The challenge that I’ve seen out there is that there are lots of offerings from both software and solutions perspectives—with everybody solving a piece of the puzzle. From a solutions perspective, one company may be able to create human models from imaging data, another might be good at assessing the structural behavior of a device, and yet another may be able to look at probabilistic assessments, and so on. But few entities can pull and synthesize all the key elements together, and do so in a rigorous manner. At Applied Science, our goal is not only to help our clients solve specific pieces of a puzzle or workflow, but also to support them in building complete end-to-end solutions that can be automated and reused efficiently. We have the expertise within TT to make a real difference for companies large and small in the life sciences world, in every step of the engineering and regulatory process.
This article was contributed by Great Ink on behalf of Thornton Tomasetti.
Subscribe to our FREE magazine, FREE email newsletters or both!
Latest News






