Connected Medical Devices Come into Their Own
The internet-enabled healthcare ecosystem encompasses sensors, devices and other systems able to acquire, process and transmit health data.

The FreeStyle Libre 3 wearable glucose sensor is just two pennies thick. The sensor is easy to apply with a one-piece applicator, and the sensor is worn on the back of the upper arm. The 14-day sensor maintains the same level of accuracy throughout the wear period. Image courtesy of Abbott.
Latest News
August 4, 2022
Healthcare digitalization is gaining momentum, due to technological advances, market trends and the current health crisis. What started with the linking of large, hardwired, hospital-based diagnostic systems has blossomed into a constellation of connected medical devices called the Internet of Medical Things (IoMT).
This new healthcare ecosystem consists of internet-enabled health sensors, devices and other systems that can acquire, process and transmit health data. The convergence of these digital technologies with the need to provide healthcare in new ways has made connected medical devices the very centerpiece of many point-of-care activities, in some cases triggering a migration of these medical devices from doctors’ offices and healthcare facilities into the patient’s home. This transformation promises to improve healthcare outcomes, expand access to care, promote operational efficiencies and enhance patient and provider experiences.
To accommodate changing healthcare demands, IoMT designers must address a new set of requirements and harness advances in computing, sensing and wireless technologies. They must do this all with an eye toward improving healthcare and providing product development teams with feedback that will help teams improve the next generation of connected medical devices.
Even as the IoMT matures and takes shape, however, questions arise concerning the future form of the healthcare ecosystem, how to overcome any challenges and finding the most effective way to manage and distribute the data generated by these systems.
An Ever-Expanding Healthcare Ecosystem
A number of factors are driving the ascent of the IoMT. Perhaps the most influential catalyst is the recent spate of technological developments.
“Advances in wearable sensors, fast and secure networks, and hybrid cloud computing, together with increased acceptance of cloud in healthcare, are enabling a major transformation in the way patients are managed across the care continuum,” says Roshy J. Francis, chief technology officer, life care solutions, at GE Healthcare. “We have already witnessed major changes in consumer health and wellness with mature wearable technologies like Fitbit and Apple Watch. This trend will continue into higher acuity care settings.”
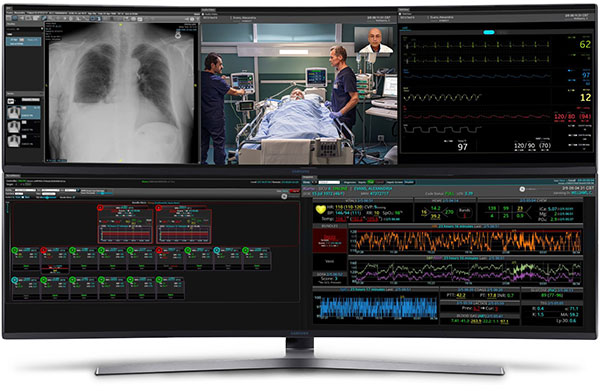
By harnessing the power of these technologies, connected medical device developers can expand the presence of the systems in the marketplace. These systems span a wide variety of device types and technologies to serve a broad spectrum of healthcare use cases. In fact, the very definition of what the devices are and the extent of the services that they deliver is in constant flux.
Some more prominent connected medical devices include the following:
- Smart consumer-grade wearables, such as Fitbit and other fitness and activity trackers and Apple watches
- Medical-grade wearables, which are used under the guidance of a clinician to manage pain or improve physical performance
- Remote patient monitoring devices, which reside in the homes of patients undergoing long-term care to help manage chronic diseases
- Smart pills, which wirelessly transmit data to medical providers regarding the patient’s internal conditions
- Point-of-care devices and systems, such as blood glucose meters, which can obtain diagnostic information and other health data without full laboratory support.
With their ability to remotely analyze and transmit data on a wide range of health conditions, IoMT systems significantly affect how chronic illnesses are monitored and treated. An example of this can be seen in the changing face of diabetes management.
“Approximately four million people are using FreeStyle Libre sensors to manage their diabetes,” says Marc Taub, divisional vice president for technical operations at Abbott Diabetes Care. “These tiny devices—which are only the size of two stacked pennies—are worn on the back of the arm and continuously measure a person’s glucose levels, sending the data every minute, automatically, to their smartphone. The glucose levels are then sent to the cloud, where they can be viewed by their healthcare team or caregiver in real time”.
In addition to these technological developments, the COVID-19 pandemic and workforce shortages have also driven IoMT technology adoption. The realities of quarantine and stay-at-home orders have dramatically accelerated trends like telemedicine and telehealth, which lay the groundwork for broader use of connected medical devices in the home.
As IoMT technology and applications mature, the influence of telehealth and telemedicine will continue to grow. This in turn will open the door for more timely, thorough and accurate diagnoses and care and increase cost savings for consumers and healthcare providers alike.
A Move Toward Open Connectivity
Though the IoMT begins with a device that measures specific physical conditions of the patient, these systems don’t really achieve their full potential until their data is transmitted to other systems. As IoMT communication technology evolves, so do the core subsystems that enable data collection and sharing.
For example, initially small or personal IoMT devices transmitted sensor data to proprietary receivers. More often, users are leveraging regulated applications, which allow them to view sensor readings directly on their smartphones.
As a result, connected medical devices increasingly use industry-standard communication protocols. Depending on the type of sensor, use environment and purpose for which it is used, various connectivity technologies may be used. Bluetooth, Wi-Fi, Medical Body Area Network and wired LAN connectivity are typically encountered and will soon expand to include 5G.
In this way, IoMT systems not only leverage the near ubiquity and convenience of smartphones, but they also leverage their cellular and network connections to transmit information to cloud-based data management systems as well, supporting remote patient monitoring.
“So, for medical images and fleet management, cable-bound internet connections are king,” says Carsten Spies, head of research and design at Siemens Healthineers. “For patient monitoring at home, we can integrate pretty much any device or wearable as long as it uses standard internet protocols.”
Software’s Significance
Against the backdrop of advances in sensing and connectivity, the healthcare industry is also witnessing an increase in the importance of software, similar to what is seen in other markets and industries.
The data analysis product is a key software element of IoMT. This application consumes any data the sensing device provides and converts it into actionable and reliable insights. Data products perform such tasks as summarizing a patient’s daily activity or identifying sleep quality trends, offering insights and recommendations.
“An example of a data product functioning in an acute care setting would be CareStation Insights Live, an application that allows an anesthesiologist to remotely view case data from anesthesia machines across multiple operating rooms on a mobile phone,” says Francis. “This offers near real-time visibility to anesthesia delivery and patient status, and it also provides the ability to select alarms displayed for important parameters.”
The quality of the software, however, is critical, and it can mean the difference between success and failure of the connected medical device.
“A poorly created software platform, or the lack of one, can create data silos,” says Francis. “A well-architectured, composable solution will facilitate better data integration, security and data privacy. A highly composable system provides components that can be selected and assembled in various combinations to satisfy specific user requirements. The high degree of composability enables unprecedented flexibility and business agility.”
Overcoming Physical and Performance Limitations
Despite the advantages that technological advances provide, connected medical device designers still face challenges. These fall into three general categories: usability, power management and upgradeability.
Because many devices are tailored for personal use, designers must reduce device form factors to facilitate mobility and wearability, while promoting usability. This is particularly true of sensors that monitor medical conditions.
For example, continuous glucose monitors lose much of their value if they are not convenient to use. Achieving the optimum form factor, however, can often be hindered by attempts to meet other requirements, such as power efficiency.
“Continuous glucose sensors must be designed to be small and easily applied,” says Taub. “The required wireless communications, however, can pose significant design challenges in terms of both the power requirements necessary to support communications over the wear duration of the device, as well as body shadowing, where signal attenuation due to the person wearing the sensor can result in the reduction in signal.”
These two challenges require careful design of the sensor’s electronics to optimize power management and communication range in a small and convenient form factor. This is made more challenging when commercial smartphones are used because there can be considerable variation in the antenna designs and communication capabilities between models.
“Take, for example, NFC [near-field communication], which has become standard on most smartphones,” says Taub. “NFC is used to activate Abbott’s FreeStyle Libre sensors, as well as to transmit glucose levels in some of the earlier generations of continuous glucose monitors. NFC antenna placement varies widely between phone manufacturers and models, such that users may need to be instructed on how to best position their smartphone during communication with the sensor.”

Developers must also ensure that connected medical device designs provide intuitive operation, easy maintenance and are inconspicuous and comfortable to wear. The system’s software should also provide easy-to-understand results and clear recommendations for follow-up actions.
Connected medical devices must also be upgradeable to stay current with frequent technology and standards changes. Complementing the upgradeability functionality—which is often implemented via remote communications—designs must also be easy to maintain.
“Device providers should be able to remotely diagnose and repair these devices when the data stops flowing due to various failure points in the complex data path between the patient and where the information is presented,” says Francis. “The large number of devices spread geographically across many settings in the future will make it difficult to manage them one issue and one device at a time, with a traditional repair person attempting to correct them.”
Privacy and Security Vigilance
Cybersecurity and patient privacy protection also continue to be a top priority. This means that IoMT systems must follow secure-by-design principles and associated processes rigorously.
“Medical device cybersecurity must be considered at every step of the device lifecycle, from ensuring that the device is delivered to the end user free of any malware, up through careful design and consideration for every potential communication interface with the device, as well as to protections of the data that have been generated from those devices,” says Taub. “This requires using layers of authentication and encryption, ensuring that hardware and software components are monitored, assessing any vulnerabilities and applying patches if required.”
A major part of this approach requires strict compliance with all relevant standards.
“Information security standards—such as ISO 27001, U.S. FDA guidance on medical device security, U.S. National Institute of Standards and Technology and U.S. Department of Health and Human Services guidelines—are resources that designers and providers deploying these solutions must use to create policies, processes and technical and administrative controls to ensure that the solutions remain cybersecure and meet applicable privacy regulations like HIPAA and data protection laws such as Europe’s General Data Protection Regulation,” says Francis.
Adherence to privacy and security standards and regulations, however, is no easy task.
“Medical device manufacturers must manage through complicated privacy laws that vary country by country and are constantly evolving,” says Taub. “This creates a very challenging environment, where compliance is driven by regional regulations that often include requirements on data residency and localization.”
Improving Future Designs
Connected medical devices bring clear benefits to patient care. But how can the data generated by these devices help to improve the design of next-generation devices and enhance the maintenance of those devices already operating in the field?
The sheer scale and complexity of these tasks is daunting, and the software platforms that manage IoMT devices are still works in progress.
“A major challenge is big data and defining scalable, cost-efficient, lossless strategies to analyze data along the pipeline, from raw sensor data to taking action based on meaningful insights, ranging widely from immediate patient intervention, to more passively scheduling an event for service, to sparking an idea for a future product improvement,” says James B. Thompson, senior director of industry strategy, medical devices and pharmaceuticals at Siemens Digital Industries Software.
One way to handle these data management tasks is to use the data mesh approach. These principles allow the clinical data to be managed in scalable, decentralized ways, which enable democratization of data. This allows various teams to contribute to and consume multiple sources of data across all domains. This opens the door for improvements of health outcomes and refinements of future device and system designs.
“The machine data—data about the device itself—provides valuable insights into the device’s performance,” says Francis. “When paired with applications that allow proactive and reactive maintenance of the device, the IoMT ecosystem can thrive, keeping cybersecure, current and reliable throughout its lifecycle. Furthermore, the data generated by the connected devices, when collected and contextualized, enables an advanced level of ‘digital empathy’ and continuous improvement of the end user and patient experience.”
Subscribe to our FREE magazine, FREE email newsletters or both!
Latest News






