Fast App: COMSOL’s Multiphysics Enables Mixing of Polymers for Medical Implants and Streamlines Manufacturing Process
When Continuum Blue's client ran into challenges in stabilizing the molding process, modeling helped uncovered the cause and take the implant to clinical trials.
Latest News
April 26, 2010
By Dr. Mark Yeoman, Continuum Blue Ltd.
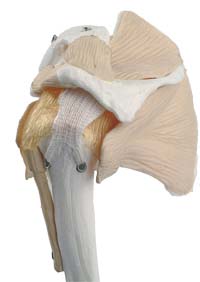 Figure 1. A fabric-based LARS implant, located in a shoulder joint, illustrating two screws through fixation holes that anchor the implant to the bone at one end of the implant. Continuum Blue is now working on multi-elastomeric version of this implant that will biodegrade once the joint has been healed. (Courtesy of LARS, France.) |
Many soft tissue medical implants are required for only a limited time, until the body’s own tissue regenerates and heals. At that point, it’s sometimes desirable to remove them, however, this isn’t always possible.
Such medical implants have traditionally been made of fabrics or biotextiles. With the advancement of special polymers, however, there is a growing movement to use hyper-elastic polymeric materials to produce implants that duplicate or augment the natural response of body tissues. Engineers at Continuum Blue Ltd. (UK) are advancing that process with the development of soft tissue implants made of biodegradable hyper-elastic elastomers that dissolve naturally in the body, eliminating the need for surgical removal.
Founded in 2004, Continuum Blue specializes in the research, development, and analysis of medical devices and implants. It has worked with several international companies, including Medtronic, Abbott Spine (now Zimmer), Synthes, NuVasive, Scient’X, British Technology Group, Ranier Technology, Aesculap, and Blackstone Medical. The company focuses primarily on the orthopedic and cardiovascular markets.
Mixing Polymers
Novel new ligament implants hold together and support bones while providing flexibility. However, they require anisotropic material properties to address various requirements. For instance, in a rotator cuff Ligament Replacement and Augmentation System (LARS) (Figure 1), the implant must be flexible enough to ensure unrestricted arm movement while providing sufficient stability to the arm and shoulder to allow the patient to support or hold an object steady. The problem is that a single elastomeric material cannot meet the desirable requirements of such a LARS.
To address this problem, Continuum Blue assisted with the development of a novel method of injecting two slow-curing polymers into a mold to create a one-piece implant with the desired anisotropic hyper-elastic properties. With a COMSOL Multiphysics model, the company’s engineers were able to determine how to best manufacture the 3D implantable device in a single production process. With this newfound knowledge, Continuum Blue has helped take a product developed with this process to clinical trials. The next step is to add a third polymer that will add the desired biodegradable properties and enhance the device’s anisotropic hyper- elasticity.
Such polymer-based LARSs have many benefits. They’re relatively inexpensive and quicker to produce than textile-based LARSs, which require multiple manufacturing steps. Additionally, there is human intervention at each of the manufacturing steps, which requires extensive quality assurance (QA). In contrast, the polymeric implant requires just two steps—the molding and the cleaning of the final product, thereby slashing manufacturing and QA costs.
The first prototypes involved two elastomers that flowed into the mold in the right concentration, sequence, speed, and condition. Each end of the molded product consists of 100% of one of the elastomers, and the regions in between have a continuous mix of the two. The company’s engineers must control the point of injection, as well as the temperatures of the injected polymers, the overall mold, and the faces of the mold. Other aspects must be controlled when the polymer enters the mold and the combined volumes.
Determining the proper parameters for all these variables is no easy task. In fact, when developing an implant of this type, Continuum Blue’s client encountered challenges in stabilizing the molding process and could not identify the cause. The company turned to Continuum Blue and simulation to gain more insight into the process, and in the end, only COMSOL Multiphysics was up to the task. In particular, the ability to handle full 3D dual-polymer injection and to control the injection profile was unique to the software and was unavailable even in the specialized injection-molding software alternatives the company evaluated.
The Model
The model itself consists of three domains: a solid region for the mold cavity walls, a liquid region for the injected polymers, and a gas region for the air in the cavity. The co-injected polymers are mixed prior to entering the mold, which is simulated by including a boundary condition that describes the volume fractions of the two polymer solutions as a function of the injection rate. This description was very easy to define in COMSOL Multiphysics.
To handle the complex interactions of the polymers with each other and the surrounding environment, Continuum Blue coupled three transient multiphysics application modes, or physics descriptions. First, a two-phase flow-phase field application mode simulates the liquid flow front as it evacuates the air from the mold cavity. Second, a phase-flow application mode simulates the two injected polymer solutions in the liquid phase of the two-phase flow and their interactions. Third, the convection and conduction application mode models the thermal changes. When the injection process stops, the filled cavity cools down. During the process, the two polymers’ densities and viscosities change, and continued fluid-fluid interaction motion occurs until high viscosities are reached and all flow stops.
The COMSOL model itself consists of roughly 50,000 elements and almost 300,000 degrees of freedom. It solves in 18 hours on a 2.8 GHz Intel Core 2 quad processor, with 8 GB of memory, running on Windows Vista.
Temperatures and Flows
Validation of the model was performed on a different complex 3D body and cavity that cannot be fully disclosed in this article. However, Continuum Blue validated its model in three ways. First, the company’s engineers performed a qualitative evaluation in which they compared a video of the liquid-air flow front during the filling process with an animated visualization of the same process in COMSOL. They found that the COMSOL model was very good at showing the fluid-air interface and the observed flow around various 3D features (e.g., walls and curved surfaces, including flow baffles) in the mold cavity at different injection rates.
Next, the engineers placed thermocouples inside the model at three locations to measure the temperature of the polymer as it flowed into the cavity and cooled. The thermocouples were located at the top, bottom, and side of the mold cavity. The readings from these thermocouples were compared with the simulation. Figure 2 shows a very good fit to the physical data. Discontinuities in the physical injection profile are due to the switching of the solenoids that control the polymer flow into the mold. In contrast, the COMSOL model uses an ideal smooth flow function. For Continuum Blue’s purposes, the thermal responses show excellent resemblance to the physical results.
Third, once the mold cooled and the polymer cured, engineers made sectional cuts in the device, looked at the boundary between the two polymer regions, and compare them with the model. Once again, the model gave a good estimation of the actual makeup of the implant (Figure 3).
Benefits
With the model, Continuum Blue can now investigate many mold variations in a reasonable timeframe and at an acceptable cost. The company estimates that a single mold redesign and trial run with the COMSOL model takes roughly 1.5 days and costs approximately GBP 850. In contrast, to do a physical sample run, where a specialized silicone mold costs approximately GBP 3000, the estimated total costs are almost GBP 9000, and the time to get the redesigned silicone mold is between three and four weeks.
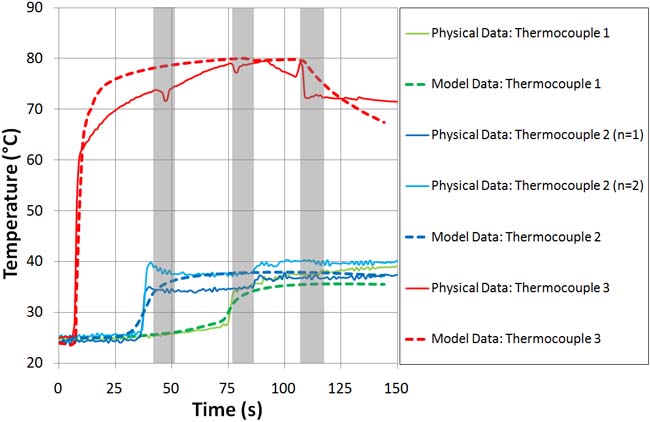 Figure 2. The left image shows a comparison of temperature readings inside the mold cavity during the filling process compared with model results. The solid lines represent measured data, and the dotted lines indicate the model data. The erratic sections in the measured data (represented by the grey regions in the graph) are due to opening and closing solenoids to control the flow of the injected polymers. The right image shows a thermal plot of the mold cavity during filling. (Courtesy of Continuum Blue.) |
Without COMSOL, finding the critical parameters that control polymer location would have taken much longer and would have cost roughly 20 times more. In addition, due to the opaque nature of the molded implant samples, engineers could not easily define the polymer boundaries and graduated regions of the physical samples (as shown in Figure 5). To do so, microbubbles or dyes had to be added to the polymer solutions, changing the polymers’ flow characteristics. Another more expensive option was to use mechanical-indent analysis to map physical material differences at points throughout the mold.
In contrast the COMSOL model gave far better insight into what was happening during the mold filling process, where the client could easily visualize the end product and the mixed regions between the two polymers. Having the COMSOL model and the resulting visualizations provide clear cost- and time- efficient benefits when convincing customers of the best mold process to provide a viable polymeric LARS solution.
Adding Biodegradability
With the validated model in hand, Continuum Blue has many projects in mind. First, it wants to increase the number of injected polymers to allow for a biodegradable product. It also wants to allow for multiple injection points.
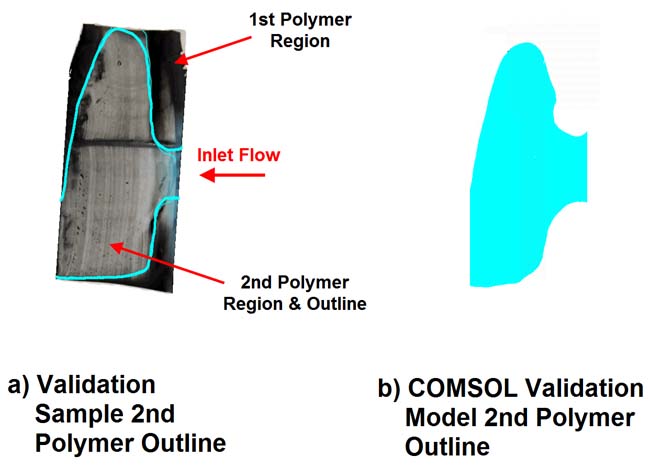 Figure 3. A cross-section from a validation sample compared with COMSOL results (blue line) shows that the outline of the second polymer matches very closely. The model shows a partial view of a 3D device different from the actual rotator cuff LARS. (Courtesy of Continuum Blue.) |
Most ambitious of all, the company wants to add parameterization to the model so that the software can help it quickly find the process parameters for new products. It will be necessary to change the size and shape of a LARS implant to account for different anatomy, joint complications, and disease. Continuum Blue hopes to create an optimized model whereby it need only describe the end product’s geometry, and the software will suggest the best mold design, along with the process parameters that will result in the desired physical properties. Hopefully, the model will cater to any mold and material design change the company’s engineers encounter.
Summary
COMSOL Multiphysics has been very valuable to finding a solution to this project. Its capabilities will certainly be beneficial and a key component to Continuum Blue’s future.
Acknowledgements
The author would like to thank Robert Snell at Ranier Technology for his assistance and help with this work
More Info
COMSOL
Subscribe to our FREE magazine, FREE email newsletters or both!
Latest News
About the Author
DE’s editors contribute news and new product announcements to Digital Engineering.
Press releases may be sent to them via [email protected].






