Materials: 3D Printing’s Medical Miracle

Oxford Performance Materials’ polymer-based cranial implants, made with AM technology from EOS, offer improved patient outcomes with reduced surgical costs. Image courtesy of Fred Smith Associates.
Latest News
October 1, 2015
 Oxford Performance Materials’ polymer-based cranial implants, made with AM technology from EOS, offer improved patient outcomes with reduced surgical costs. Image courtesy of Fred Smith Associates.
Oxford Performance Materials’ polymer-based cranial implants, made with AM technology from EOS, offer improved patient outcomes with reduced surgical costs. Image courtesy of Fred Smith Associates.A groundbreaking 3D-printed splint has been credited with restoring a baby’s breathing. Cranial implants used to patch missing portions of the skull are being 3D printed to fit a patient’s exact anatomy, and 3D-printed biomaterial scaffolds are growing human tissue that will eventually help to regenerate bone and other anatomy.
Such medical miracles are happening at a more frequent pace thanks to major advances in the world of 3D printing, from new and improved bio-compatible materials to the evolution of software and printing processes that promise to make good on the era of personalized medicine.
Well-known materials such as titanium, stainless steel, and a variety of polymers and ceramics have already gotten the FDA stamp of approval for use in 3D printed medical applications where bio-compatibility is crucial, including implants, surgical guides and instrumentation. There is also plenty of R&D in the area of material science, exploring new compounds and formulations that will have a major impact on future 3D printing medical applications.
In the near term, however, much of the emphasis is on modifying and enhancing existing 3D printer technologies to exploit the wide mix of available materials. Companies like Stratasys, 3D Systems, Autodesk, Materialise and EOS, in tandem with a range of other companies and researchers, are devising new printing processes and software that can open doors to new 3D printing medical applications.
“All 3D printing companies are developing completely new materials, but trying to get those approved for medical use is a lengthy and costly process,” says Peter Mercelis, Ph.D., director, Applied Technologies, Healthcare, for 3D Systems. “Therefore, the [current] challenge for 3D printing companies is to be able to process well-known, validated materials with completely new manufacturing technology.”
Mix and Match Materials
For years, commonly used alloys have been used to produce fully dense metallic parts for medical applications using traditional manufacturing processes, but it wasn’t until about five years ago that was possible to do the same with 3D printing, Mercelis says. “The Direct Metal Printing (DMP) printers that existed 10 years ago were not powerful enough to process those materials and achieve fully-functional parts,” he says. Over the years, 3D Systems, along with other 3D printer manufacturers, have evolved their offerings to incorporate more powerful lasers capable of higher heat intensity, enhanced optical components, and new machine architectures so they can produce metallic parts that can live in the body for long periods of time and don’t pose any risk of contamination.
The next phase has been to figure out how to alter the properties of existing materials, creating whole new classes of options that didn’t exist before. By tuning the printer hardware and with the introduction of new software, new material combinations and the ability to design and print intricate lattice structures are enabling researchers and practicing physicians to create hip implants, face implants and other structures that are cell friendly, thus can stimulate growth of human tissue.With its digital materials strategy, Stratasys, for example, now offers over 1,200 materials for use with select PolyJet printer models. Users can combine two or three PolyJet base resins in specific concentrations and structures to create a composite material with the desired properties. Its Fused Deposition Modeling (FDM) printer lines offer three material choices that are FDA approved, including the ULTEM 1010 material, which is high-temp rated so it can withstand the sterilization practices mandated in hospitals, says Scott Rader, general manager for Medical Solutions at Stratasys.
“There really haven’t been any fundamental physics hurdles to get over—it’s really about looking at the applications and understanding the needs of the customer,” Mercelis says. Stratasys delivers a range of bio-compatible materials with various strength, durability, textural and cost properties to support such applications as 3D-printed medical models, surgical tools and implants that can left in the body on a semi-permanent or permanent basis, he says. “When looking at your materials choices, it’s important to have a solution for all three use cases,” Rader says.
Moving forward, Stratasys is pouring lots of effort into refining its printing practices to take advantage of already known materials to expand the range of medical applications—for example, refining a high-strength plastic that could replace steel used in orthopedic implants. “It’s not fundamentally about discovering new materials, it’s about learning how to adapt 3D printing processes to make use of known biocompatible materials,” says Rader—for instance, modifying 3D printers to be capable of the higher temperatures required to use some of the high-strength plastics.
 Along with a biocompatible rating, Stratasys ULTEM 1010 resin boasts high heat resistance, chemical resistance and tensile strength. Image courtesy of Stratasys.
Along with a biocompatible rating, Stratasys ULTEM 1010 resin boasts high heat resistance, chemical resistance and tensile strength. Image courtesy of Stratasys.Researchers and physicians at the University of Michigan are at the forefront of such 3D printing modification efforts. A team there has adapted a biopolymer called polycaprolactone (PCL) for use on a Direct Laser Sintering (DLS) printer from EOS to create a reabsorbable trachea splint used to fix a collapsed bronchus that was preventing a baby from breathing. The team worked with a materials vendor Oxford Performance Materials to adapt PCL into a powdered mix form that could be used with the EOS P100 DLS 3D printer. It also developed software, used in tandem with the Mimics modeling program from Materialise, to design the trachea implant from the patient’s own scan data so it was an exact match to the baby’s anatomy, explains Dr. Scott Hollister, Ph.D., professor of Biomedical Engineering and Mechanical Engineering and associate professor of Surgery at University of Michigan.
The University of Michigan team has done three more similar procedures and is exploring use of other thermoplastic materials such as PEEK and its polymorphous cousin PEKK, which supports many different applications and a variety of customized materials from the same base molecule. The materials selected for this application all promote cell regeneration, Hollister explains, and can be reabsorbed by the body, ensuring that once human tissue forms, the scaffolding dissolves away.
The advent of such materials is making 3D printing invaluable for such regenerative medical applications, Hollister says. “Our anatomy design has complicated shapes and porous structures that can’t be built with existing methods like injection molding,” he says. “We’re just beginning to see 3D printing’s impact on enabling the design of implants specifically made for the individual patient.”
The University of Michigan team was able to push the envelope because it could modify the EOS DLS printer to work with thermoplastic materials thanks to EOS’ open architecture strategy, according to Andy Snow, senior vice president of EOS North America. “They were able to create their own materials to process in our system … because we give people the ability to adjust various parameters in our machine, whether it’s the heating platform, thickness or exposure strategies,” he says. “That’s an important part of taking medical applications to the next level. You have to show flexibility.”
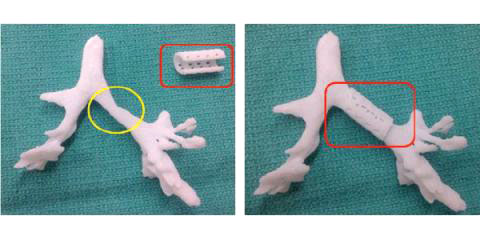 A University of Michigan team developed a biodegradable scaffolding and tracheal splint. Image courtesy of University of Michigan.
A University of Michigan team developed a biodegradable scaffolding and tracheal splint. Image courtesy of University of Michigan.In addition to facilitating the design of personalized implants, 3D printing can also be far more cost effective than traditional manufacturing processes like injection molding because it eliminates the amount of secondary processes required. “If you want to put a porous structure on an implant in the traditional way, you have to use foam paste, spread it on and then use a secondary heat treatment process, which can be very expensive,” says Snow. “[With 3D printing], you can design the porous structure as part of the CAD file, eliminating the secondary processes and saving time and ultimately, saving costs.”
Adding Lattice Structures
Creating the complex lattice structures used as a basis for implant design has been an on-going challenge that is now starting to be addressed in earnest. In addition to the work being done at University of Michigan and other research facilities, a number of well-known software companies are working on this problem, including Autodesk. Autodesk recently announced Autodesk Within Medical, a generative design software program designed to help biomedical engineers create micro lattice porous structures for custom orthopedic implants. The software, built with an optimization engine, takes input parameters such as desired weight and maximum stress to generate micron-accurate designs that incorporate “variable-density lattice structures and surface skins” that promote fixation of bone and encourage cell growth, officials say.
 Printed on Autodesk’s Ember DLP printer, the Zygoma bone is filled with a trabecular lattice and printed with PEG (Polyethylene glycol) material. Image courtesy of Autodesk.
Printed on Autodesk’s Ember DLP printer, the Zygoma bone is filled with a trabecular lattice and printed with PEG (Polyethylene glycol) material. Image courtesy of Autodesk.While most of the current implants designed in Within Medical are being printed in metal, Autodesk’s bio/nano team is researching the use of other organic materials for use with the software. The team is conducting its research on a modified Autodesk Ember, a high-resolution DLP (Direct Light Processing) printer built on the company’s open source Spark 3D printing platform.
One such research initiative involves using the modified Ember 3D printer, Within Medical software, and new biomaterials to design a trabecular lattice structure that is lighter weight than traditional solid titanium implants and predisposed to promote cell growth.
“We are trying to leverage the flexibility of the Ember printing to do 3D printing of materials never done before,” says Larry Peck, senior director, Autodesk Bio/Nano Research Group. “We are applying the Within design software to achieve higher resolution on lattice that’s amenable to cell growth. By stitching together different technologies, we can come up with solutions we have never had before.”
More Info
Subscribe to our FREE magazine, FREE email newsletters or both!
Latest News
About the Author
Beth Stackpole is a contributing editor to Digital Engineering. Send e-mail about this article to [email protected].
Follow DE





